Device therapy is increasingly employed in the management of complex congenital heart disease (CHD). Bradycardias, most often related to sinus nodal dysfunction (SND) or atrioventricular nodal (AVN) block, may necessitate the implantation of pacing devices, while malignant arrhythmias may be treated by appropriate use of implantable cardioverter defibrillators (ICDs). However, there is a complex interplay between these classical device indications and associated supraventricular tachyarrhythmias, failure of ventricular function and ventricular dyssynchrony. These considerations, alongside the technical demands of implantation in the context of complex anatomy, necessitate a tailored and nuanced approach in the use of device therapy in the complex CHD population.
This review focuses on patients with CHD of moderate and severe complexity, including those with single ventricle circulation and transposition of the great arteries.1 The management of congenital complete heart block is not addressed directly in this review, but shares many of the management principles discussed for the paediatric group.
Pacing Devices in Complex CHD
Guidelines and Prevalence of Pacing Indications
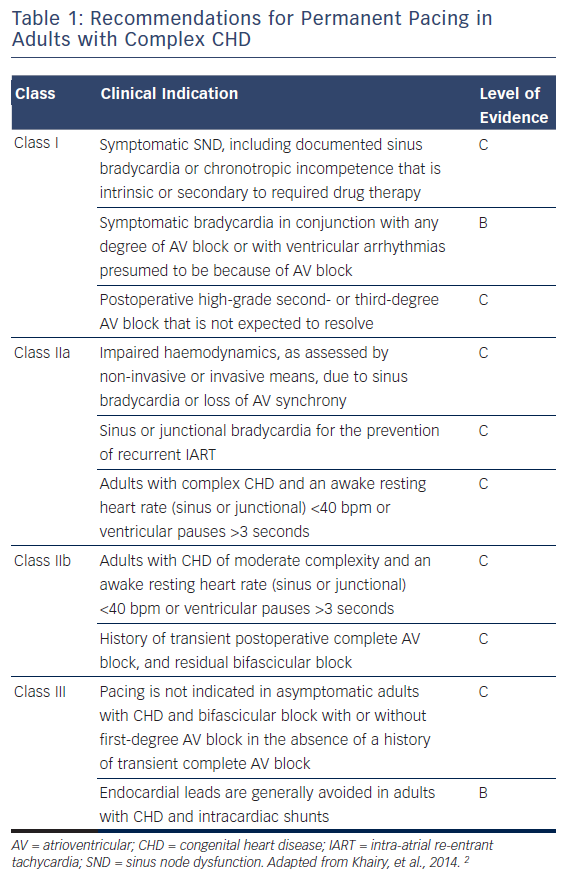
Guidelines for pacing in the complex CHD cohort are limited by a lack of prospective randomised trials, and the marked heterogeneity of the group. The indications for permanent cardiac pacing in adult patients with complex CHD are summarised in Table 1, but in children the majority of prospective data relate to patients with congenital complete heart block, and practices are generally extrapolated from those principles.
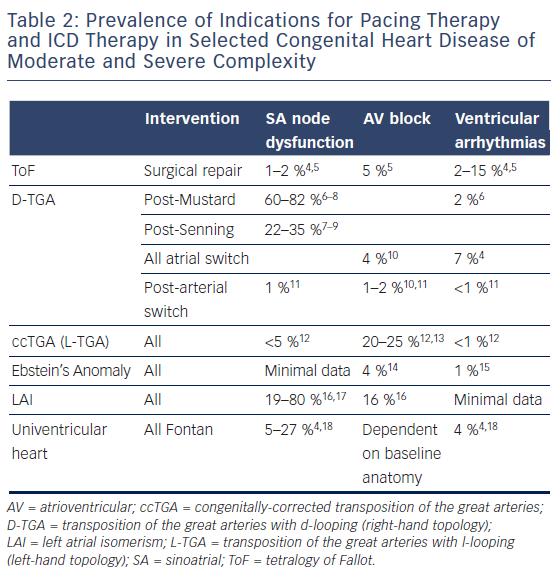
The incidence of sinus nodal and atrioventricular (AV) nodal dysfunction in the more common complex CHD lesions is summarised in Table 2. It is clear that a significant proportion of these patients will require pacing, in addition to those with intra-atrial conduction block and His- Purkinje disease. Sinoatrial and AV nodal dysfunction may be congenital in conditions such as left atrial isomerism (LAI) and congenitallycorrected transposition of the great arteries (ccTGA; L-transposition), but more frequently the cause is iatrogenic following corrective or palliative surgery.
It is also important to note the association of indications for pacing with the increased incidence of atrial and ventricular tachycardias.3 SND, AV dyssynchrony and ventricular bradycardia predispose to the triggering of tachyarrhythmias, and the common pathogeneses of abnormal conduction, surgical scar and progressive myocardial fibrosis are evident. Therefore, device therapy should not be considered in isolation, and a multimodal approach is necessary, combining antiarrhythmic medication, catheter ablation and anti-tachycardia pacemaker (ATP) therapies.2
Endocardial versus Epicardial Pacing and Number of Leads
Epicardial pacing systems possess obvious advantages for patients with complex CHD. They do not rely upon venous access, may be placed during concurrent surgery, avoid endovascular leads and associated thromboembolic events19 and present fewer concerns regarding lead extraction. However, these factors need to be weighed up against increased lead failure rates and pacing thresholds, with a consequent reduction in system longevity.14,19,20 In a retrospective study of 287 patients with CHD, Silvetti et al. found a clear survival benefit of transvenous leads (10-year lead survival 71 % versus 95 %, p<0.0001).20 Steroid eluting leads are improving the efficacy of epicardial leads, and the potentially life-threatening implications of lead failure are mitigated to some degree by newer technologies such as transtelephonic monitoring and auto capture algorithms, providing a degree of safety during the early failure period. However, the decision for endocardial versus epicardial pacing should be made on a case-by-case basis.
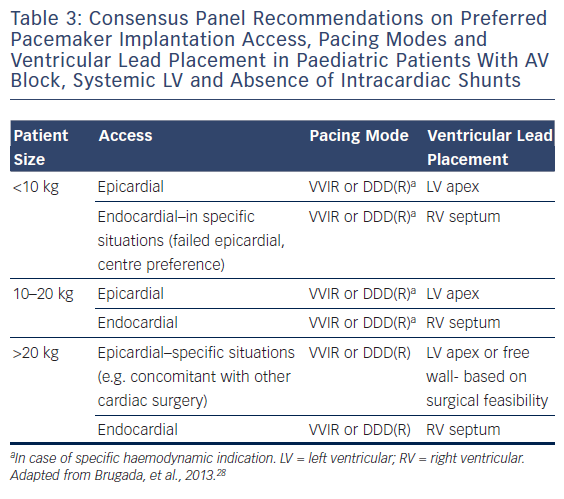
For the smallest patients, epicardial systems are generally recommended (see Table 3), and minimally invasive epicardial pacemaker approaches are also now being employed at some centres.21 However, selected patients <10 kg may continue to be treated with an endovascular system.22
For those in whom an endovascular approach is most appropriate, a comprehensive evaluation of shunts (or potential shunts) by echocardiography or angiography should be undertaken before lead implantation (Class 1C recommendation),3 in order to evaluate for thromboembolic risk. Thin lumenless pacing leads (model 3830 SelectSecure®, Medtronic Inc.) may reduce complications in smaller patients and those with limited venous lumens. These 4.1 Fr leads were first introduced in 2004 and are catheter delivered: conventionally using an 8.4 Fr delivery system, but the use of smaller 5 Fr sheaths has also been reported. They have demonstrated a good electrical performance and low complication rate,23 and may be more straightforward to extract than conventional leads despite the absence of a lumen for locking stylet.24
In addition, it should be borne in mind that generally fewer leads implanted will result in fewer complications. For children with AV block and normal ventricular function, VVIR may be the pacing mode of choice. DDD pacing with transvenous leads is technically feasible in small children, but may not be needed.25 There are also other situations in which the benefit of additional leads are outweighed by risk. As for patients with structurally normal hearts, single chamber AAI(R) pacing is frequently indicated for the large cohort of CHD patients with SND. However, those with difficult access to the ventricle, such as Fontan circulation, also stand to derive minimal overall benefit from an additional ventricular lead if the indication is marginal (see below).
Device selection should also include the requirement for future magnetic resonance (MR) imaging. MR conditional pacing systems are now being widely adopted, with growing evidence of safety in a 1.5T MR environment,26 but artefact within the field of view has a substantial impact upon image quality.27 Therefore pre-emptive MR imaging prior to implantation should be considered, particularly for functional and scar assessment.
Lead Positioning
Pacing site selection is now dictated by a multitude of factors, and simply adequate sensing and pacing thresholds are no longer sufficient. Directable delivery sheaths and active fixation leads mean that most endocardial targets are accessible, while epicardial lead positioning has also been shown to be increasingly important in determining long-term outcome.29 Preservation of ventricular function should be taken into consideration in all decisions for lead placement.
In general, atrial lead placement is less critical, but atrial septal pacing may reduce unnecessary ventricular pacing when compared with appendage pacing.30 Ventricular lead placement is highly dependent upon the cardiac anatomy and whether an endocardial or epicardial approach has been adopted. For endocardial pacing in the AV concordant biventricular heart, there is some evidence for optimal site selection. Pacing from the right ventricle (RV) apex results in marginally better left ventricle (LV) ejection fraction than non-RV apical sites (e.g. right ventricular outflow tract, mid-septum and upperseptum) 29 and this finding is replicated on systematic review.31 It is also clear that RV free wall pacing is inferior.29,32 Further lead-implant guidance techniques that include evaluation of ventricular function, via pressure-volume loops or the first derivative of systolic pressure (dP/ dt), may also optimise chronic paced ventricular function and have demonstrated that there is no single optimal site for endovascular lead placement.33,34
In studies of epicardial lead placement, Janousek et al. performed a non-randomised retrospective trial of lead placement in 178 children undergoing permanent pacing, with structurally normal heart, and there was a demonstrated benefit to placement of the epicardial ventricular lead at the LV apex or midlateral wall.29 The only prospective study of LV apex or free-wall pacing was conducted in neonates and infants with congenital complete AVB and showed good ventricular systolic function and synchrony after one year of pacing.35 Trials in complex CHD are generally lacking, and the evidence base for optimal lead positioning is small, but again the RV free wall should be avoided if possible.
Resynchronisation Therapy
Cardiac resynchronisation therapy (CRT) has been shown to be a useful therapy for adult patients with selected forms of electrical dyssynchrony and heart failure with reduced ejection fraction, but patient selection and technical issues create significant challenges in the setting of CHD.36 Most cases of CRT in the paediatric and CHD population do not have left bundle branch block, but instead it is performed as an upgrade to conventional ventricular pacing because of pacing-associated heart failure (>60 % of cases), or with a failing systemic RV.37 In recent guidelines, indications for CRT in CHD are generally Class IIa and IIb, but should not be considered in adults with narrow QRS complex (<120 ms) or life expectancy <1 year (Class III).2
Assessing Dyssynchrony and CRT indications
Electrical activation delay, with bundle branch block on the side of the systemic ventricle (left bundle branch block for systemic LV, right bundle branch block for systemic RV), identifies the group of patients that are most likely to respond to CRT.32 However, the further assessment and definition of dyssynchrony in many forms of repaired CHD are controversial. Mechanical dyssynchrony, as assessed by echocardiography, is the most widely used measure, but there are no universally adopted or validated selection criteria in CHD.37 As well as conventional echocardiographic modalities, strain assessment, speckle tracking and 3-dimensional volume assessment of both the LV and RV are likely to play a significant role in assessment for CRT in the future.38,39 It should also be noted that, within the normal cardiac anatomy population, the use of echocardiography measures of dyssynchrony to select patients for CRT has not been found to be robust, with a lack of reproducibility from centre-to-centre and also little evidence that much is added beyond traditional measures of QRS duration and morphology.40 Non-echocardiographic parameters, such as those derived from cardiac MR imaging or electrocardiographic imaging,41 may also play a role.
Overall, the efficacy of CRT in CHD is likely to rely upon a multitude of factors, and identifying potential responders and non-responders will be highly dependent upon anatomy and valvular function as much as systolic function and dyssynchrony alone. However, objective assessment will become an increasingly pressing issue, with a recent review by the European Heart Rhythm Association recommending dyssynchrony assessment and consideration of CRT in a wide range of patients.28
Technical Challenges and Outcomes from Resynchronisation
There are often significant technical challenges in achieving CRT in complex CHD, particularly via the endocardial route. Even if the coronary sinus is accessible, pacing of the systemic ventricle may not be achievable via this route, for example following atrial switch for TGA with d-looping, i.e. right-hand topology (both the venous baffle and the coronary sinus lead to the LV, with no access to the RV). Access may also be complicated by surgical repairs and baffles, and therefore an epicardial or hybrid approach may be required.39
Outcome data for CRT in CHD are generally limited to small studies in the acute postoperative setting, or longer term follow-up studies with a variety of surrogate endpoints (6-minute walk test, hospitalisations, quality of life, ventricular stroke volume/ejection fraction). In general, the systemic LV is more likely to respond to CRT than the systemic RV,42 and patients listed for transplant may respond to a sufficient degree to enable delisting.43 In multisite pacing of the single ventricle, with widely spaced electrodes, the benefit is reduced but there are still reports of improvement.42,44 A recent study from Osaka, evaluating the outcome in patients with a systemic RV, has suggested that the leads should be placed at furthest sites in the longitudinal RV direction, unless there is substantial short axis dyssynchrony, in which case the leads are best placed laterally on opposite sides of the ventricles.45 Further CRT optimisation, including refinement of AV and VV (interventricular) delay, may also be considered, but there is an absence of evidence in the CHD population for the optimisation guidance modality and settings.
Programming of pacing devices
Beyond the decision for single, dual or multisite pacing, there are also considerations for pacing programming. The general principle of minimising ventricular pacing applies in CHD as much as the general population, and novel algorithms have been developed by major manufacturers that incorporate a strategy to allow extended AV delays, and thus reduce ventricular pacing (Boston Scientific: AV search hysteresis; St Jude: autointrinsic conduction search). Medtronic Inc. have employed an alternative technique (managed ventricular pacing) that incorporates a mode-switching strategy between AAI(R) and backup ventricular/DDD(R) pacing with loss of AV conduction.39 Recent consensus guidelines on pacing in CHD generally class minimisation of ventricular pacing as a Class IB recommendation.2 In addition, further basic programming principles should be considered in the CHD population, including limiting upper tracking rate in patients at risk of intra-atrial re-entrant tachycardia (IART).
Strong consideration should also be given for implantation of systems capable of administering ATP therapies, particularly in those with a documented history of IART. A dual chamber system is generally required for atrial ATP, and a recent study of 80 implants in patients with CHD found ATP to be successful in terminating 57 % of IARTs, with high success rates in atrial switch patients.46 In patients with a single ventricle where an endocardial ventricular lead cannot be placed, we plug the ventricular port to give an open circuit on the ventricular channel allowing effective ATP.
Implantable Cardioverter Defibrillators in Complex CHD
Indications and Guidelines
There is now a reasonable body of literature that details the risk of sudden cardiac death in complex CHD, and the subsequent indications for ICD implantation. Implantation after a cardiac arrest or sustained spontaneous ventricular tachycardia (secondary prevention) is generally a Class IB indication for ICD implantation in CHD, where no reversible cause is identified.2,3,28,47
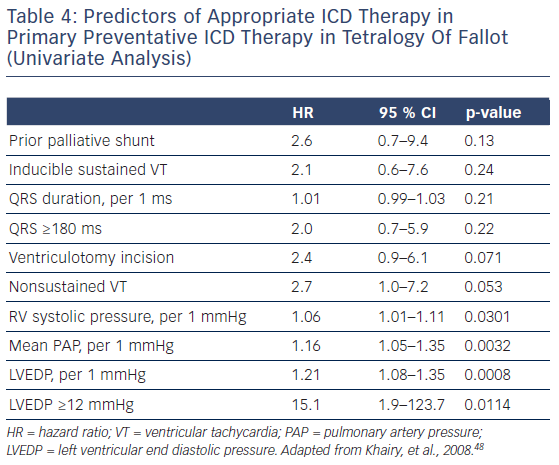
However, evidence for selection for primary preventative therapy is limited in the complex CHD population.36 The most detailed data relate to those with repaired tetralogy of Fallot (ToF). A raised left ventricular end diastolic pressure, pulmonary artery pressure and RV systolic pressure are the strongest predictors of appropriate shock therapy. The significance of inducible sustained ventricular tachycardia at electrophysiology study is unclear, with a trend towards increased incidence of appropriate shocks in inducible patients,48,49 but it is symptomatic non-sustained ventricular tachycardia that is the more important prognostic indicator.49 An annual appropriate shock rate for primary preventative ICDs in ToF has been reported to be as high as 7.7 % but with an inappropriate shock rate of 5.8 % per year and other system complications occurring in nearly 30 %.48 The ‘appropriate shock rate’ may also include transient non-haemodynamically disturbing arrhythmias that may not have needed treatment.
Outside the ToF population, the indications are less well delineated, reflected in a significantly lower appropriate shock rate in the adult population.50 Conventional non-CHD indications may be applicable for those with biventricular circulation,2 but the systemic RV systolic function cut-off value that defines benefit is likely to be lower than the 35 % for the LV.51 The largest study of ICD therapy in CHD is that of Berul et al. in 2008.52 In a review of 443 patients (69 % of whom had structural heart disease), 48 % were for primary prevention. Of these, 18 % received appropriate shocks but 20 % received inappropriate shocks. The impact of an ICD in the generally younger CHD population should not be underestimated, with the likelihood of multiple generator and lead replacements, other complications and psychosocial stressors related to ICD therapy. It is also suggested that the inappropriate shock rate is increased in young and active CHD patients, aggravating concerns of lead fracture, particularly in those with Fidelis leads (Medtronic Inc.).52,53
Devices and Access
Issues regarding access for ICD therapy are similar to those for conventional pacing, but complicated by larger lead sizes. Patients with univentricular hearts or post-atrial switch may also benefit from ICD therapy but pose further access difficulties,39,51,54 and pericardial, pleural or subcutaneous lead placement of the standard transvenous coils should be considered55,56 combined with endocardial or epicardial sensing leads. In addition, there may be a role for individualised calculation of optimal generator placement site, based upon the cardiac anatomy, and early work has been performed in computing ICD configuration based on cardiac MR-derived anatomy.57
Subcutaneous ICD
There is increasing evidence for the use of subcutaneous ICD (S-ICD; Boston Scientific), avoiding the potential intravascular and lead complications of other systems. They are, however, relatively bulky and do not allow for conventional pacing or ATP, and therefore may generally be more appropriate for channelopathies and other arrhythmias not associated with structural CHD. Early evidence of efficacy and complications is beginning to emerge, with relatively promising results.58–61 Initial concerns regarding sensing issues, inappropriate shocks and failure of conversion of malignant arrhythmias seem to have been broadly overcome with appropriate programming and placement, but S-ICD remains a technology in its early phases with very little evidence in the structural CHD population.59 However, its implementation in patient groups with no venous access to the heart, particularly after a Fontan operation, is attractive if pacing is not required.
ICD Programming Considerations
Inappropriate shock rates are relatively high, largely related to supraventricular arrhythmias, T-wave oversensing and increased lead failure rate.53 As with all ICDs, the task of balancing overtreatment against delaying therapy is a delicate one. The relatively slow atrial arrhythmias, predisposing to 1:1 conduction, may necessitate the implementation of manufacturer-specific discriminator algorithms, such as PR Logic® (Medtronic Inc.), morphology and onset analysis. These should be used with caution, however, as supraventricular tachycardia characteristics may vary widely from episode to episode.62 Faster cut-off rates, longer detection times and customised ATP sequences should also be considered.
Considerations for Devices in Complex CHD
The implanting physician must have a detailed understanding of the patient’s cardiac and central vascular anatomy, alongside the prior procedures and diagnostic reports. Further cross-sectional imaging and assessment of vascular patency may often be required in patients who have undergone multiple prior procedures. In addition, haemodynamic concerns such as residual shunts or baffle patency should also be carefully considered prior to implantation. The need for concomitant or subsequent interventional or surgical procedures, as part of the overall management of the patient, must also be weighed up prior to device implantation.
Single Ventricle Circulations
Superior Cavo-pulmonary Connections
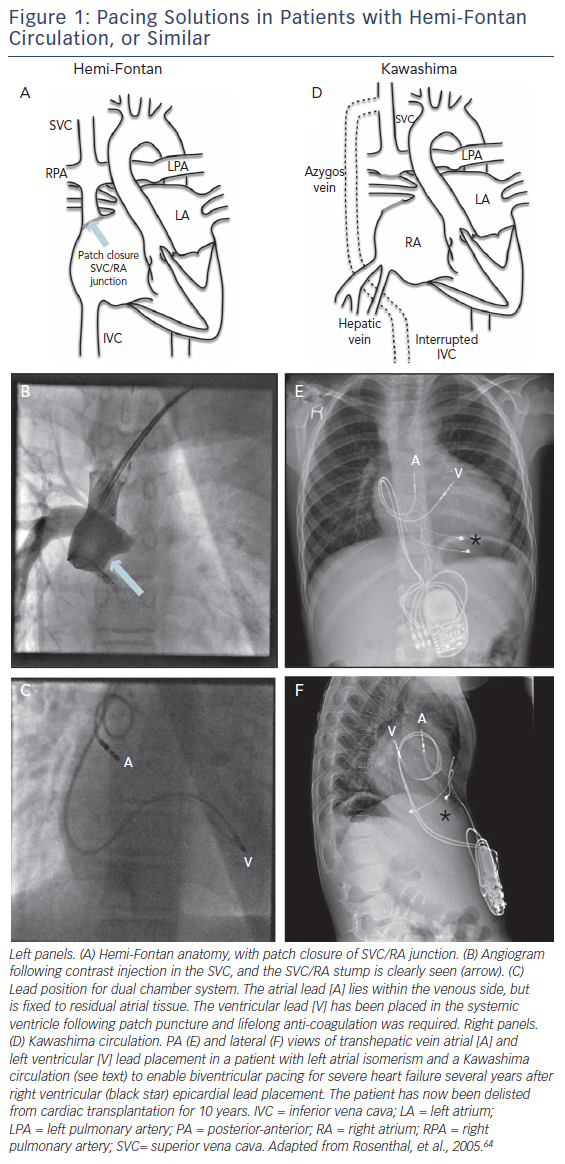
One and one-half ventricle circulations, with hemi-Fontan or Glenn, may preclude conventional placement of the atrial lead. However, in the hemi-Fontan there is frequently atrial tissue remaining on the pulmonary artery side of the patch, and successful long-term pacing has been reported (Figure 1).63 The Glenn anastamosis tends not to have any residual atrial tissue on the pulmonary side, and in this case puncture from the pulmonary artery to atrial mass would have to be considered (see below). The Kawashima circulation, in which venous drainage from the lower half of the body is to the superior vena cava via the azygos vein with interrupted IVC, represents a further complication (Figure 1: D). This arrangement is most commonly encountered in patients with LAI, who are at markedly increased risk of complete heart block. Unfortunately, access to the ventricle in particular can be extremely challenging, and transhepatic access may be required,64 unless the technique of Arif et al. is used (Figure 2E and F).66
Total Cavopulmonary Circulation
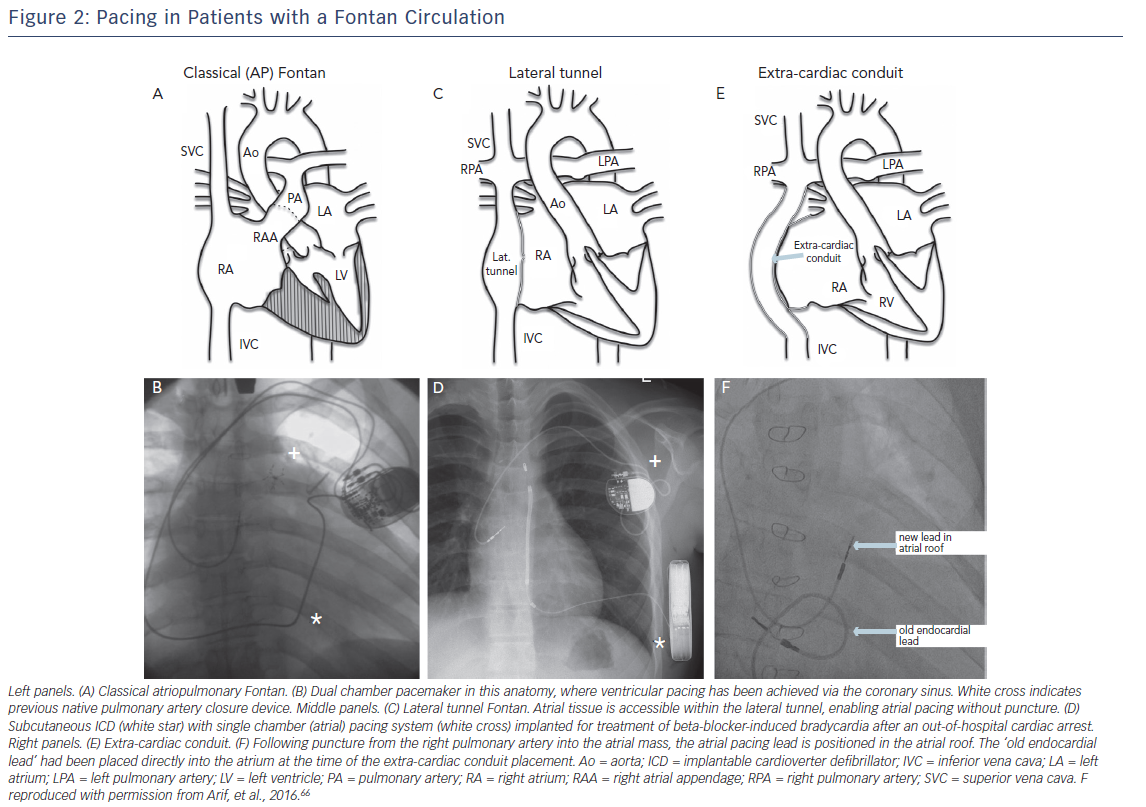
Atrial pacing in the classic atriopulmonary Fontan and lateral tunnel Fontan generally poses relatively few problems in terms of access to atrial myocardium. In those with extra-cardiac conduit epicardial pacing is generally required. However, transpulmonary artery pacing may be an alternative, puncturing through the inferior aspect of the left65 or right66 pulmonary artery into the common atrium (Figure 2F).
Ventricular pacing in most Fontan anatomies is more easily achieved via epicardial leads, but multiple prior surgical procedures often render this approach unattractive and other endovascular techniques may be considered. For patients with atriopulmonary Fontan, and occasionally lateral tunnel Fontan, the coronary sinus may be accessed from the venous side (Figure 2B).67 Alternatively, the lead may be placed endocardially via a fenestration or baffle puncture, but endocardial lead placement evaulation should be made taking into account the substantial thromboembolic risk,19 and we would always anticoagulate these patients.
ICD therapy in Fontan patients is often best served by non-transvenous systems (epicardial or subcutaneous). The use of a conventional subcutaneous system is limited by the absence of pacing capabilities, and may necessitate two devices (Figure 2D). As an alternative, shock coils may be placed in the pericardial or pleural space, with the ICD generator usually placed abdominally. The non-transvenous ICD system is likely to have significantly shorter longeivity than transvenous,55 but circumvents many of the potentially severe complications and may still have a role to play in the Fontan or hemi-Fontan patient, even with the advent of licensed subcutanous ICD systems.28
Atrial Switch Procedures
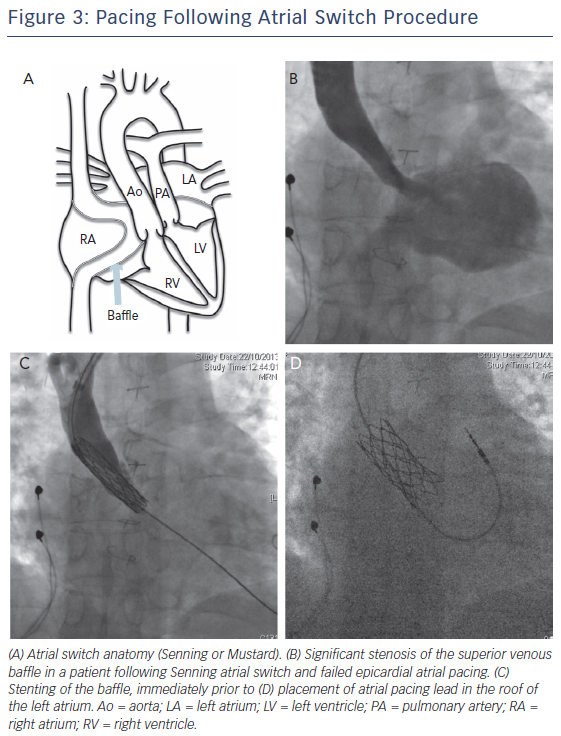
Atrial lead placement following the atrial switch procedure should be performed bearing in mind that the baffle tends to direct the pacing lead to the lateral wall of the LA, close to the left phrenic nerve (Figure 3). In addition, there is a high incidence of systemic venous obstruction, particularly of the superior limb following the Mustard procedure (>40 % of patients)8 and consideration should be given to stenting of the baffle, if required, prior to lead insertion. Baffle leaks are also highly prevalent, and systemic anticoagulation or baffle leak occlusion may be required.19
The ventricular lead placement is to the smooth LV, and active lead fixation has greatly improved pacing success. The lead should ideally be placed on the septum to avoid pacing the left ventricular free wall that may cause diaphragmatic pacing and dyssynchrony. However, resynchronisation therapy cannot be performed via a coronary sinus lead, as the default ventricular lead position is already within the LV. CRT is therefore likely to require a hybrid epicardialendocardial approach, or baffle pucture with the long term risk of placement of a pacing lead within the systemic ventricle. The use of RF energy to perforate the baffle and enable lead placement in the systemic RV has been described, for both CRT-pacing and CRT-defibrillator therapy. Access was generally via the subclavian approach with transoesophageal echocardiographic guidance, with good haemodynamic results.68
For those undergoing primary ICD placement, a relatively high complication rate (27 %) and low appropriate shock rate has been reported. Careful risk stratification is therefore required before implantation, particularly in view of the bulky leads that must cross the superior venous baffle.69
Device-Related Complications and Lead Extraction
Complications of Permanent Pacing in CHD
Permanent cardiac pacing in CHD is linked with a higher rate of pacing complications than in non-CHD. In a review of epicardial and endocardial pacing in CHD by McLeod et al.,14 it was found that the overall long-term complication rate in CHD was close to 40 %, compared with 5 % (or 0.5 % per year) in non-CHD. Epicardial systems were linked to lead failure, particularly exit block and lead fracture, whilst endocardial systems were more durable but susceptible to dislodgement and insulation leaks. A study of the paediatric population by Fortescue et al.70 made similar findings of high incidence of lead failures in all groups, but suggested a general move towards transvenous pacing where possible.
In those that have undergone endocardial pacing, though, in addition to the spectre of lead extraction, two further potential complications should also be considered: venous occlusion and thromboembolic events. Venous occlusion is generally accepted to be a significant risk in smaller patients, but its incidence is dependent upon patient and lead size and difficult to define: a figure of around 5–10 % has been quoted at long-term follow-up.19,71 Regarding thromboembolic events in patients with an intracardiac shunt, the risk is more than doubled in patients with endocardial systems from 0.6 % per year (in patients with no permanent pacemaker or an epicardial system) to around 2 % per year.19
Lead Extraction
The need for lead extraction remains a major concern for those placing systems in younger patients and in systemic chambers, and the risks and benefits need to be weighed up at the time of potential extraction. Conventional lead extraction guidelines also apply to the complex CHD population,2,72 but the benefit of extraction of non-functioning leads is likely to be greater in younger patients.
There are generally limited data on lead extraction specifically in the complex CHD population. A retrospective study by McCanta et al. (2013)73 reviewed 24 laser lead extractions (LLE) in CHD patients, compared with matched non-CHD patients. They found a significantly lower extraction success rate in the CHD cohort (74 % versus 92 %) using LLE, but many of the remaining leads were then removed using other techniques, and there were no significant complications. On the other hand, Cecchin et al.74 found no significant difference between the CHD and non-CHD patients, using a combination of simple extraction with a non-locking stylet with complex techniques such as the radiofrequency-powered sheath when required, as did Khairy et al. using LLE.75
Five-year View
Device therapy in complex CHD has always necessitated ingenious, creative and sometimes brave approaches to adapt pacemakers and defibrillators that have been designed for the normal adult heart. Smaller leads and devices have assisted the progression of the field in the paediatric group, but there are further future innovations that promise to be highly applicable for those with complex CHD.
There are a number of solutions that are close on the horizon. These include drug-eluting coatings for pacemakers to reduce infections, advanced imaging processing to guide planning and implantation, and a growing understanding of the capabilities of telemonitoring and how to harness the strengths of the technology.
Leadless pacemakers have already been licensed in Europe for implantation in the RV, and are close to FDA approval. However, device size, extraction concerns and lack of dual chamber capabilities need to be resolved before extensive use in the complex CHD population. The combination of a leadless atrial device with subcutaneous-ICD, though, may be closer on the horizon and could prove invaluable if ATP is available.
Conclusion
Device placement in the patient with complex CHD is not an isolated procedure, and should be viewed in the context of the patient as a whole. Multidisciplinary discussion prior to implantation is almost invariably required, with input from congenital cardiac surgeons, interventionalists, imaging specialists and electrophysiologists informing a balanced approach with appropriate long-term considerations. The heterogeneity of the patient group means that almost every implantation decision requires tailoring to the patient. The evidence base for any individual approach, be it epicardial versus endocardial, dual chamber versus biventricular pacing, or pacing lead removal versus retention, is difficult to apply and there is no substitute for a detailed understanding of the patient anatomy, prior procedures and device indications. The need for concomitant or subsequent interventional or surgical procedures as part of the overall management of the patient may tailor the approach more substantially than device considerations alone.