Atrial fibrillation (AF) is the most common sustained arrhythmia in adults and confers increased risk of death,1 thromboembolism and impaired quality of life.2 Current pharmacological antiarrhythmic drugs provide limited protection against AF recurrence and have poor safety profiles, while invasive ablation treatments are associated with significant risks and limited long-term success rates. Moreover, neither of these treatments has been documented to reduce adverse outcomes associated with AF.3,4 Several pathophysiological processes have been proved to be involved in AF, such as inflammation, oxidative stress, endothelial dysfunction, initiating triggers (often from pulmonary veins), changes in autonomic tone in addition to fibrosis and ischaemia. Prevention of AF with interventions that modify these substrates or target specific mechanisms for AF has emerged as a result of recent experimental studies suggesting the use of upstream therapies. Non-antiarrhythmic drugs that have been tested in prevention of AF include angiotensin-converting-enzyme inhibitors, angiotensin receptor blockers, statins and long chain-3 polyunsaturated fatty acids (n-3 PUFA). n-3 PUFA have multiple effects on cardiac electrophysiology,5–10 such as membrane stabilisation in the myocardial cell by prolonged inactivation of the fast sodium outward channel, resulting in a longer refractory time and provide protection from ventricular arrhythmias and sudden death,11–13 but also have antifibrotic, anti-inflammatory and antioxidant characteristics that might influence the mechanisms involved in the initiation and maintenance of AF.14
In several experimental animal models, pre-treatment with n-3 PUFA decreased the development and progression of atrial fibrosis, reduced the abundance of collagen in atrial appendage and the duration of induced episodes of AF.15,16 They also prevented, in this kind of model, significant shortening of the atrial effective refractory period associated with AF, reduced inducibility of AF and sustainability of induced AF and attenuated structural changes in the atrial myocardium.16
Results of epidemiological studies have been controversial: In the Cardiovascular Health Study, the consumption of boiled or baked fish one to four times per week was associated with a 30 % lower risk of incident AF at 12 years compared with fish consumption less than once a week.17 However, in other population-based studies no association was found between n-3 PUFA intake and incident AF. Both the Danish Study and the Physicians’ Health Study showed that the patients with higher fish intake were more likely to develop AF: in the Danish study adjusting hazard ratios (HRs) for incident AF at 5.7 years, in quintiles 2–5, were 0.86, 1.08, 1.01 and 1.34 (p for trend = 0.006) compared with the lowest quintile and in the Physicians’ Health Study patients with the highest fish intake (≥5 meals per week) were more likely to develop AF compared with those eating fish <1 time per month (RR 1.46; 95 % CI 0.94–2.28).18,19
Finally, no association between development of AF at 3 years and fish intake was found in the Women’s Health Initiative study, which was carried out in more than 45,000 women.20 All these studies were based on food frequency questionnaires to assess fish intake and none provided data on serum n-3 PUFA content and its relationship with development of AF. In the Kuopio Ischaemic Heart Disease Risk Factors Study, designed to investigate risk factors for cardiovascular diseases, atherosclerosis and related outcomes in middle-aged men from eastern Finland, the serum concentration of n-3 PUFA was measured in 2,174 men. Only high docosahexanaeoic (DHA) acid but not eicosapentaenoic (EPA) acid content was associated with reduced risk of incident AF (HR 0.62; 95 % CI 0.42–0.92; p=0.02) suggesting that the preventive effect may depend on the use of a specific acid.21 These results are consistent with other studies in which DHA has been shown to be able to inhibit cardiac arrhythmias in rats and to have a beneficial effect on heart rate variability in humans.22,23
In this context, the focus of several randomised clinical trials in n-3 PUFA has been on two AF populations: patient for whom the objective was to maintain normal sinus rhythm after cardioversion or spontaneous restoration of sinus rhythm and patients in whom the objective was to prevent AF after cardiac surgery. The aim of this paper is to give an up-to-date review of the results of these trials and of recently published meta-analyses on this topic, trying to explain the conflicting results that have emerged by focusing on methodological aspects and on possible pathophysiological mechanisms, such as the role of inflammation and oxidative stress, as suggested by more recent studies and from biohumoral subanalysis of these trials.
Effects of n-3 Fatty Acids on Prevention of Recurrent Atrial Fibrillation
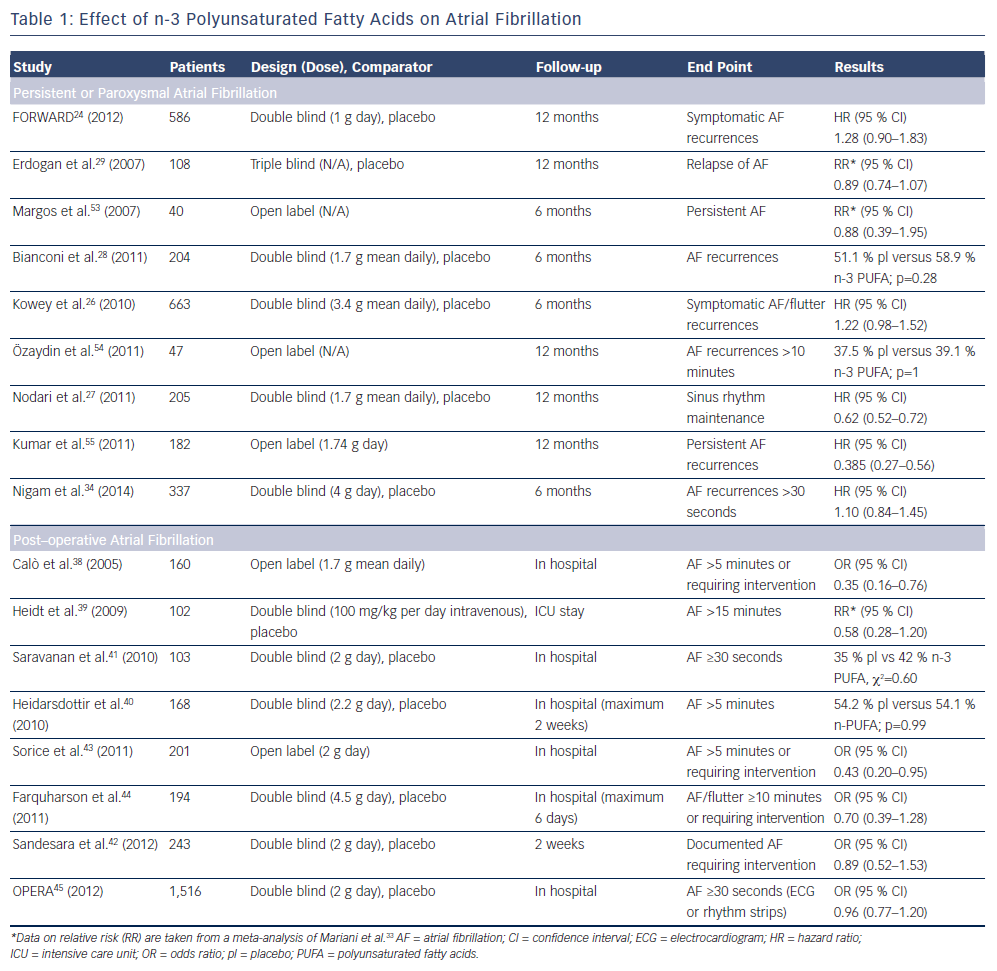
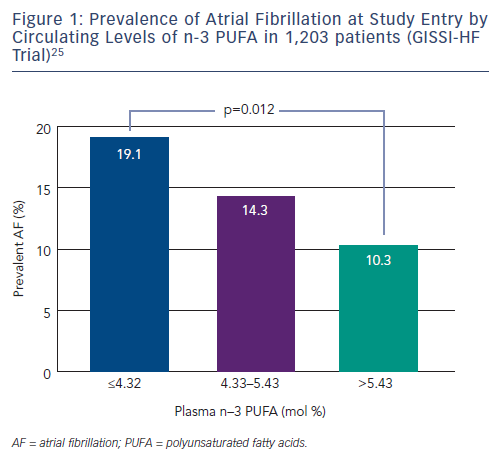
Several trials have been designed to evaluate the effect of n-3 PUFA in the prevention of AF recurrence (see Table 1). In FORWARD (Randomised Trial to Assess Efficacy of PUFA for the Maintenance of Sinus Rhythm in Persistent Atrial Fibrillation), 586 patients with previous symptomatic episodes of AF were randomised to receive 1 g/day n-3 PUFA or placebo in a double-blind fashion; the primary efficacy endpoint was the time to first recurrence of AF; the follow-up duration was 12 months. At the end of the study, 18.9 % in placebo group and 24.0 % in n-3 PUFA had recurrent AF (HR 1.28 95 % CI 0.90–1.83) and the lack of statistical differences between randomised patients to placebo or n-3 PUFA were observed for all the other study outcomes (composite of all-cause mortality, non-fatal stroke, non-fatal acute MI, systemic embolism, heart failure development and severe bleeding).24 In a post hoc analysis of the GISSI HF trial (Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico Prevenzione), the effect on new episodes of AF of n-3 PUFA daily supplementation was examined in 5,835 patients with heart failure.25 In this trial, fish consumption at study entry was estimated through a self-administered questionnaire and circulating levels of n-3 PUFA were measured in a subgroup of 1,203 patients. More frequent fish consumption with diet was associated, in univariate analysis, with a lower prevalence of AF but after the adjusted analysis fish intake was not associated with AF prevalence while, among patients with measured serum levels of n-3 PUFA, those in the lowest tertile (≤4.32 mmol %) had a twofold higher risk of AF than those in highest tertile (>5.43 mmol %) (OR 1.84; 95 % CI 1.15–2.95; p=0.012) (see Figure 1).25 During the follow-up period of 3.9 years, 15.2 % of the patients allocated to n-3 PUFA and 14.6 % of those randomised to placebo developed AF (unadjusted HR 1.10; 95 % CI 0.96–1.25; p=0.19).
The efficacy of n-3 PUFA supplementation in the prevention of AF was assessed in a randomised trial of 663 outpatients carried out in the US. Patients had symptomatic paroxysmal (542 patients) or persistent AF (121 patients) without structural heart disease and they were treated with 8 g/day of n-3 PUFA for the first 7 days and with 4 g/day thereafter for through 24 weeks. At the end of the study, there were no differences between treatment groups for recurrence of symptomatic AF in both paroxysmal (HR 1.15; 95 % CI 0.90–1.46; p=0.26) and persistent stratum (HR 1.64; 95 % CI 0.92–2.92; p=0.09).26
Several studies have been conducted in the clinical setting of postcardioversion recurrence of AF. The preliminary results of a randomised trial showed that therapy with 1 g/day of n-3 PUFA in 199 patients with recurrent persistent AF was associated with a significantly lower incidence of recurrence compared with placebo at 1 year (40 % versus 72 %; p=0.007).27 These results were not supported by the results of other randomised trials. In the study by Bianconi et al., 204 patients with persistent AF were randomised to receive 3g/day of n-3 PUFA until electrical cardioversion and 2g/day thereafter for 6 months.28 AF relapsed in 58.9 % of the n-3 PUFA and in 51.1 % of the placebo patients (p=0.28). Similarly, no favourable effects of 4 weeks’ pretreatment with n-3 PUFA on recurrence of AF after cardioversion were shown in 108 patients.29
Systematic reviews published in the last few years did not provide a definitive answer on the role of n-3 PUFA in prevention of AF probably because the number of patients was relatively small;30–32 in a recent meta-analysis including 16 studies, the analyses were conducted separately for persistent or post-operative AF, eight studies (1,990 patients) evaluated the effect of n-3 PUFA on reverted persistent or paroxysmal AF with a follow-up ranging from 6 to 12 months.33 Treatment had no effect on AF recurrence (RR 0.95; 95 % CI 0.79–1.13) and there were no statistical differences in the overall death rate (RR 0.85; 95 % CI 0.26–2.77). In 2014, a new randomised placebo controlled trial was published: 337 patients with symptomatic paroxysmal or persistent AF, documented within 6 months from enrolment, were randomised to high doses of fish oils (4 g/day) or placebo and followedup on average for 271±129 days. The primary endpoint was time to first AF recurrence lasting at least 30 second and it did not differ between groups (64 % in fish oil group versus 63 % in placebo group; p=0.5). Results were consistent across all pre-specified subgroups such as diabetic, hypertensive, paroxysmal or persistent AF patients, except for a significantly higher AF recurrence for fish oil users with ischaemic heart disease (HR 2.6; 95 % CI 1.1–6.1).34
These data, in combination with those from previous trials, do not recommend the use of n-3 PUFA to prevent recurrent or incident AF (see Table 1).
However, many secondary prevention trials had a short follow-up duration and, for this reason, important benefits from n-3 PUFA could be missed. The Vitamin D and Omega-3 Trial (VITAL) rhythm substudy is examining the impact of the administration of 1 g/day of n-3 PUFA on incident AF 25,875 men and women without cardiovascular disease over 5 years of follow up. It is possible that with a more prolonged duration of treatment a potential benefit could emerge.35
Effects of n-3 Fatty Acids on Post-operative Atrial Fibrillation Prevention
Cardiac surgery is often complicated by the occurrence of supraventricular arrhythmias and particularly AF (post-operative AF [POAF]). Almost 30–40 % of patients undergoing coronary artery bypass grafting (CABG) surgery develop POAF, most frequently between the second and the third post-operative day.36 This percentage rises nearly to 70 % in patients undergoing concomitant valve surgery. The risk of developing this arrhythmia is related to the presence of several clinical risk factors (e.g.prior history of AF, heart failure, left atrial enlargement, left ventricular systolic dysfunction, post-operative withdrawal of beta-adrenergic receptor blockers and chronic obstructive pulmonary disease), but the stronger predictor is advancing age.37 POAF has both clinical and economic implications: patients who develop POAF are at increased risk of cerebrovascular events, haemodynamic instability and congestive heart failure. Furthermore, POAF is associated with increased length of stay in the intensive care unit and prolonged hospitalisations and is therefore responsible for significant patient morbidity and healthcare costs.36
Several drugs (e.g. amiodarone, sotalol and beta-adrenergic receptor blockers) have proved effective in reducing the risk of POAF. As multiple acute factors contribute to the development of AF after cardiac surgery (enhanced sympathetic and parasympathetic tone, atrial stretch, fluid and electrolyte abnormalities, metabolic abnormalities, oxidative stress, myocardial and pericardial inflammation) other treatments have been tested and are currently under evaluation. Based on the evidence of anti-inflammatory and antioxidant proprieties of n-3 PUFA several studies have been designed to prove their efficacy on POAF prevention (see Table 1).
Calò and colleagues evaluated the efficacy of n-3 PUFA for the prevention of POAF in 160 patients randomised to polyunsaturated fatty acids (n-3 PUFA 2 g/day) and to a control group. Treatment was initiated at least 5 days preoperatively and continued until hospital discharge. Patients randomised to n-3 PUFA had a significantly lower incidence of POAF compared with the control group (15.2 % versus 33.3 %; p=0.013) and a mean shorter hospital stay (7.3 [2.1] versus 8.2 [2.6] days; p=0.017).38
Heidth et al. randomised 102 patients to n-3 PUFA (100 mg fish oil/kg body weight/day) given intravenously or free fatty acids (100 mg soya oil/kg body weight/day) starting on admission to hospital and ending at discharge from intensive care. POAF (primary endpoint) occurred in 15 patients (30.6 %) in the control and in 9 (17.3 %) in the n-3 PUFA group (p<0.05). Furthermore, patients allocated to a n-3 PUFA treatment had to be treated in the intensive care unit for a shorter period of time than the control patients.39
Based on these promising results, Heidarsdottir and colleagues conducted a prospective, double-blinded, placebo-controlled trial randomising 168 patients admitted for CABG and/or valvular repair surgery to receive n-3 PUFA capsules (1,240 mg EPA and 1,000 mg DHA) or olive oil capsules for 5–7 days prior to surgery and postoperatively until hospital discharge. Unlike previous studies, the authors found no difference in the incidence of POAF (54.2 versus 54.1 %; p=0.99) and in the length of stay.40
Similar disappointing results were found by Saravanan et al. who failed to demonstrate the efficacy of n-3 PUFA, 2 g/day administered orally (versus olive oil, placebo) in reducing the incidence of POAF in 103 patients undergoing CABG surgery. Despite higher n-3 PUFA levels in serum and right atrial tissue in the active treatment group, there was no significant difference between groups in the primary outcome of POAF (placebo versus n-3 PUFA; 43 % versus 56 %; p=0.28).41
Sandesara et al. in the Fish Oil for Reduction of Atrial Fibrillation After Cardiac Surgery (FISH) trial randomised patients undergoing CABG to pharmaceutical-grade n3 PUFAs 2 g orally twice daily (minimum of 6 g) or a matched placebo ≥24 hours before surgery. Despite a higher n-3 PUFA dose, a longer time of treatment (2 weeks after surgery) and larger sample size (260 patients) compared with previous similar studies, the rate of POAF was similar in both groups (30 % n-3 PUFAs versus 33 % placebo; p=0.67).42
Sorice and colleagues randomised 201 patients undergoing CABG to n-3 PUFA 2 g/day or placebo for at least 5 days before surgery and until hospital discharge. The authors found a significant reduction in the incidence of POAF in the n-3 PUFA group. Interestingly, subgroup analysis showed a significant reduction of POAF only in the group including patients treated with n-3 PUFA undergoing ‘on-pump’ CABG surgery, with no effect on those who underwent ‘off-pump’ cardiac surgery. Notably, therapy with n-3 PUFA had no effect on length of post-operative hospital stay.43
Farquharson and colleagues undertook a double-blind randomised controlled trial to examine the effectiveness of fish oil supplementation on POAF after CABG and valve procedures in 200 patients randomised to receive fish oil (providing 4.6 g/day of n-3 PUFA) or a control oil starting 3 weeks before surgery. The longer period of pre-treatment did not have the expected results: 194 subjects completed the study, with 47 of 97 subjects in the control group and 36 of 97 subjects in the fish oil group developing POAF in the first 6 days after surgery (primary endpoint, OR 0.63; 95 % CI 0.35–1.11). Notably there was a significant decrease in mean length of stay in the ICU in the fish oil group (ratio of means 0.71; 95 % CI 0.56–0.90).44
The conflicting results of all these previous trials gave the impulse for the organisation of an international multicentre double-blind, placebocontrolled, randomised clinical trial, the Omega-3 Fatty Acids for Prevention of Post-operative Atrial Fibrillation (OPERA) trial, with the enrolment of 1,516 patients scheduled for cardiac surgery. Patients were randomised to receive n-3 PUFA (1 g capsules containing ≥840 mg n-3 PUFA as ethyl esters) or placebo, with preoperative loading of 10 g over 3 to 5 days (or 8 g over 2 days) followed postoperatively by 2 g/day until hospital discharge or post-operative day 10, whichever came first. The primary endpoint (POAF lasting longer than 30 seconds) occurred in 233 (30.7 %) patients assigned to placebo and 227 (30.0 %) assigned to n-3 PUFAs (OR 0.96; 95 % CI 0.77–1.20; p=0.74). Furthermore, no significant differences between the two groups were observed for any of the secondary endpoints: post-operative AF lasting longer than 1 hour, resulting in symptoms, or treated with cardioversion; post-operative AF excluding atrial flutter; time to first postoperative AF; number of AF episodes per patient; hospital utilisation; and major adverse cardiovascular events, 30-day mortality. Treatment was safe in terms of bleeding and other adverse events.45
Two interesting analyses46–7 on biohumoral data from the OPERA trial have recently been published confirming the limited influence of mechanical and haemodynamic factors and reinforcing the role of oxidative stress in the pathogenesis of POAF. N-terminal of the prohormone brain natriuretic peptide (NT-proBNP) or high-sensitivity cardiac troponin T (hs-cTnT) on the morning of surgery, or changes in their levels between morning of surgery and post-surgery, were not significantly associated with POAF after adjustment for clinical and surgical characteristics.46 On the contrary a relatively linear association with incident POAF of validated, fatty acid-derived oxidative stress biomarkers (F2-isoprostanes, isofurans and F3-isoprostanes) measured in plasma and urine on the day of surgery and on the second post-operative day could be described.47
Two metanalyses,33,48 which included the same eight38–45 clinical trials discussed above, on POAF prevention with n-3 PUFA have recently been published. Interestingly, the authors obtained opposite results: Mariani et al.33 found no significant benefit, whereas Costanzo and colleagues48 found that preoperative supplementation with n-3 PUFA significantly prevented the occurrence of POAF in patients undergoing cardiac surgery. The difference in results of the two studies can be explained by the different adjudication of events in the trial of Saravanan and colleagues.41 Costanzo and colleagues48 used the primary outcome measure of any AF >30 seconds in the monitor recordings (number of events with n-3 PUFA versus control, 22 versus 18) whereas Mariani and colleagues33 considered clinical AF (29 events in the n-3 PUFA group versus 22 in the controls). Costanzo and colleagues conducted a subgroup analysis showing particular benefit in patients undergoing CABG surgery.
In accordance with the results of the biohumoral analysis in the OPERA trial,46,47 and following the hypothesis of a strong involvement of oxidative stress in the development of POAF, Rodrigo and colleagues49 designed a controlled trial to assess whether the reinforcement of the antioxidant system, through n-3 PUFA plus antioxidant vitamin supplementation, could reduce the incidence of POAF. A total of 203 patients scheduled for on-pump cardiac surgery were randomised to placebo or supplementation with n-3 PUFA (2 g/day) (EPA:DHA ratio 1:2), vitamin C (1 g/day) and vitamin E (400 IU/day). POAF occurred in 10 of 103 patients (9.7 %) in the supplemented group versus 32 of 100 patients (32 %) in the placebo group (p<0.001). The efficacy of antioxidant therapy with vitamin C in preventing POAF as well as reducing in hospital stay was confirmed in a recently published metanalysis.50 The results of trials discussed do not support the routine use of n-3 PUFA in POAF prevention, but their combination with other drugs with antioxidant proprieties seem to have promising perspectives that should be confirmed in larger adequately powered controlled trials.
Conclusions
The efficacy of n-3 PUFA in preventing AF has been tested, with conflicting results, in several randomised clinical trials conducted both in patients with a history of AF and in those undergoing cardiac surgery (the so-called POAF). The results of these trials do not support the routine use of n-3 PUFA in AF prevention in both settings. Accordingly, both European51 and US guidelines52 do not recommend their systematic use in clinical practice. The promising results of the combination of n-3 PUFA with other drugs with antioxidant proprieties need to be confirmed, providing interesting pathophysiological explanation of the conflicting results of the studies by pointing out the importance of oxidative stress over inflammation in the pathogenesis of AF and particularly of POAF.
Clinical Perspective
- Atrial fibrillation (AF) is associated with an increased risk of fatal and nonfatal events and current antiarrhythmic drugs provide limited protection against AF recurrence and have poor safety profile.
- Understanding of the pathophysiological mechanisms that underlie AF has led to the development of upstream strategies for AF prevention.
- n-3 polyunsaturated fatty acids (n-3 PUFA) are safe, well tolerated and have multiple effects on cardiac electrophysiology and both clinical and preclinical studies have shown ‘antiarrhythmic properties’.
- The efficacy of n-3 PUFA in preventing AF has been tested, with conflicting results, in several randomised clinical trials conducted both in patients with history of AF and in those undergoing cardiac surgery (so-called post-operative AF).
- The results of these trials do not support the routine use of n-3 PUFA in AF prevention, but their combination with other drugs with antioxidant proprieties seem to have promising perspectives.